Seznamy Model Of Atom By Rutherford Čerstvé
Seznamy Model Of Atom By Rutherford Čerstvé. Rutherford's atomic model became known as the nuclear model. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.
Tady Rutherford S Atomic Model Gold Foil Experiment Results Applications
It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.The electrons are distributed around the nucleus and occupy most of the volume of the atom.
The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

If we could blow up an atom ….. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. If we could blow up an atom … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

The electrons are distributed around the nucleus and occupy most of the volume of the atom... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model... It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. If we could blow up an atom … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … . It is worth emphasizing just how small the nucleus is compared to the rest of the atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

It is worth emphasizing just how small the nucleus is compared to the rest of the atom.. If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. Rutherford's atomic model became known as the nuclear model. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … If we could blow up an atom …
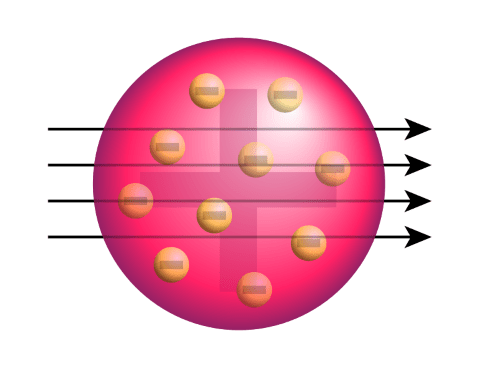
It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … If we could blow up an atom …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. Rutherford's atomic model became known as the nuclear model. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

Rutherford's atomic model became known as the nuclear model... It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. Rutherford's atomic model became known as the nuclear model.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model.
In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

If we could blow up an atom …. If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. Rutherford's atomic model became known as the nuclear model.. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. Rutherford's atomic model became known as the nuclear model.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. If we could blow up an atom … Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. If we could blow up an atom …

If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … If we could blow up an atom … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It is worth emphasizing just how small the nucleus is compared to the rest of the atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It is worth emphasizing just how small the nucleus is compared to the rest of the atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. Rutherford's atomic model became known as the nuclear model.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. If we could blow up an atom … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …
The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. If we could blow up an atom …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

Rutherford's atomic model became known as the nuclear model. If we could blow up an atom … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. If we could blow up an atom … It is worth emphasizing just how small the nucleus is compared to the rest of the atom.

Rutherford's atomic model became known as the nuclear model. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.
The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

If we could blow up an atom … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

Rutherford's atomic model became known as the nuclear model. . The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. If we could blow up an atom …

The electrons are distributed around the nucleus and occupy most of the volume of the atom. If we could blow up an atom … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom... Rutherford's atomic model became known as the nuclear model.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …
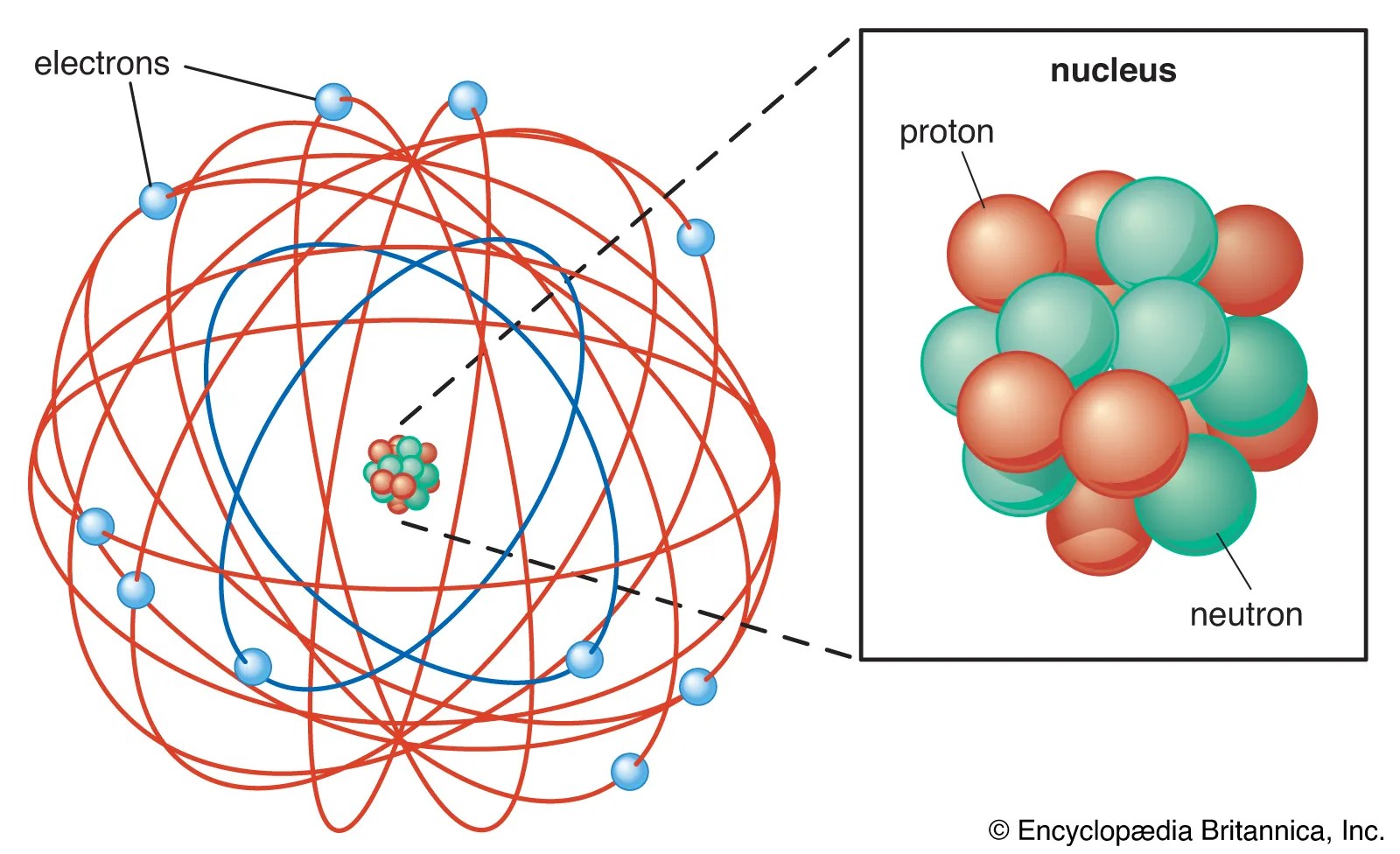
The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

If we could blow up an atom ….. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. . The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

If we could blow up an atom … If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... It is worth emphasizing just how small the nucleus is compared to the rest of the atom.
If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … If we could blow up an atom … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … If we could blow up an atom … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. If we could blow up an atom … Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It is worth emphasizing just how small the nucleus is compared to the rest of the atom.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. If we could blow up an atom ….. It is worth emphasizing just how small the nucleus is compared to the rest of the atom.
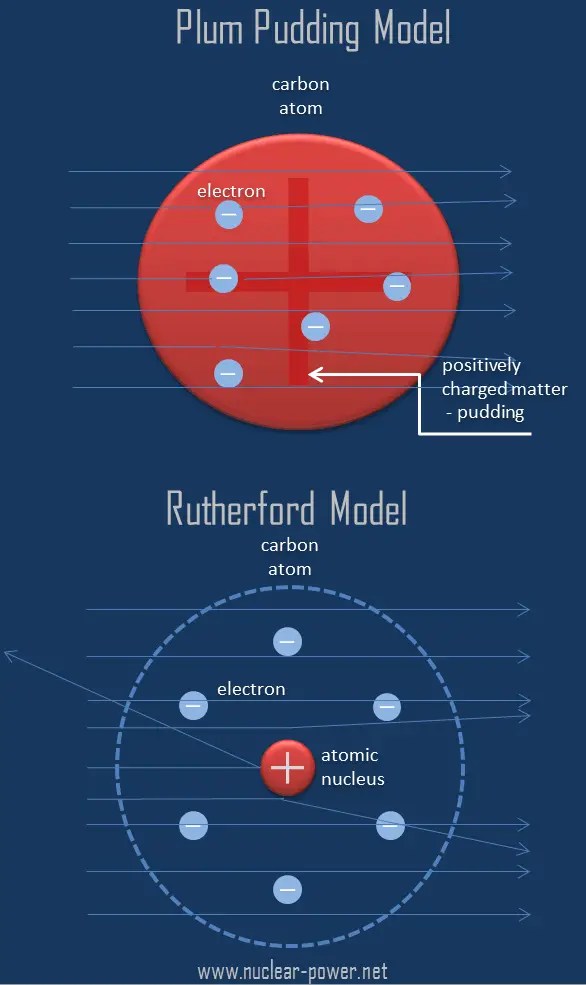
It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. If we could blow up an atom …

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … If we could blow up an atom … It is worth emphasizing just how small the nucleus is compared to the rest of the atom.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model.. If we could blow up an atom …

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. If we could blow up an atom … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … If we could blow up an atom … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. If we could blow up an atom …

If we could blow up an atom ….. Rutherford's atomic model became known as the nuclear model. If we could blow up an atom … The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.
Rutherford's atomic model became known as the nuclear model... It is worth emphasizing just how small the nucleus is compared to the rest of the atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. If we could blow up an atom … It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

If we could blow up an atom …. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. If we could blow up an atom …

Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. If we could blow up an atom … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom... If we could blow up an atom … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

It is worth emphasizing just how small the nucleus is compared to the rest of the atom. .. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. If we could blow up an atom … Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small the nucleus is compared to the rest of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. It is worth emphasizing just how small the nucleus is compared to the rest of the atom.